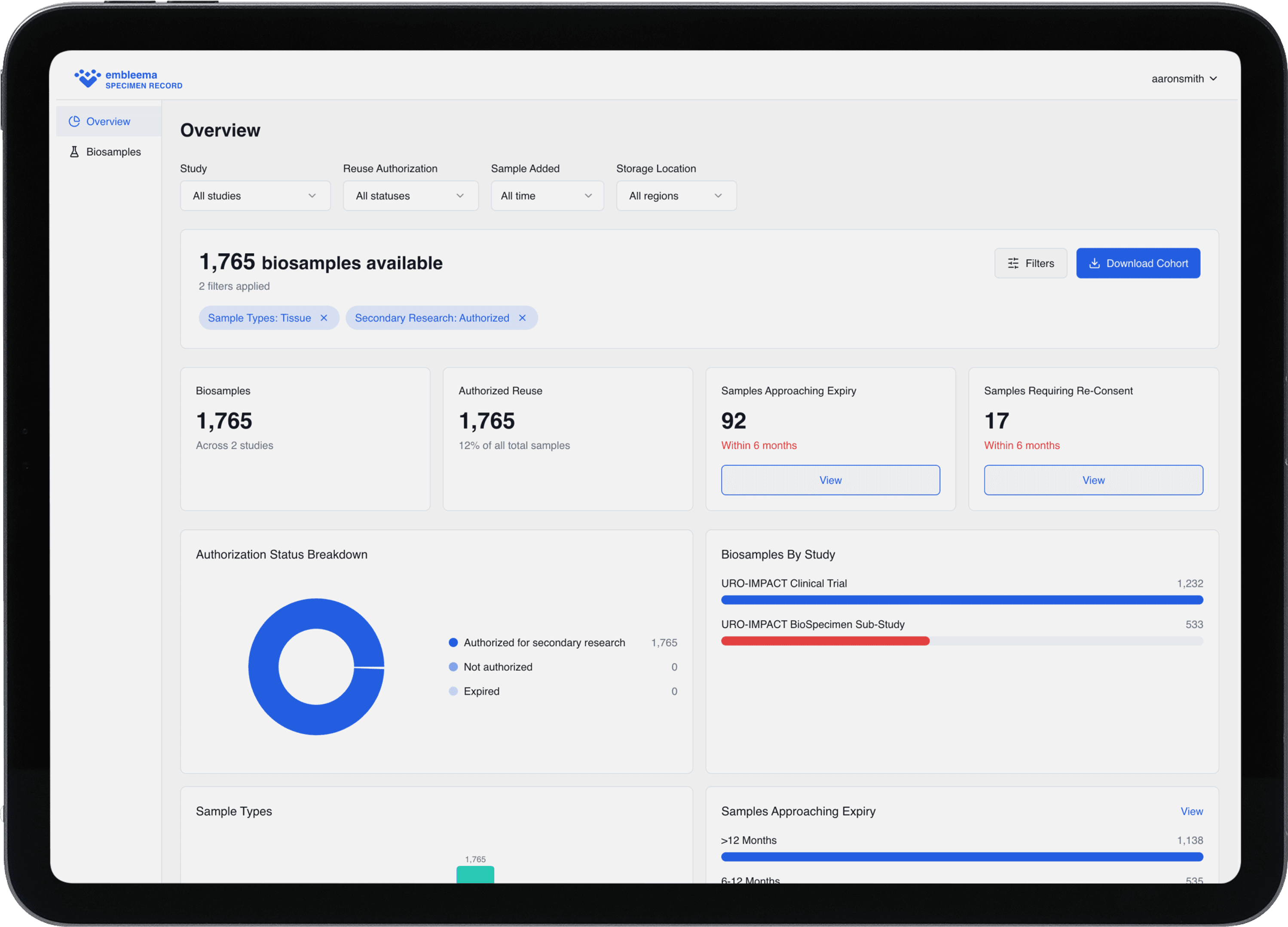
The operational gaps limiting secondary use
Consent uncertainty
Unclear authorization for secondary research use creates compliance risks.
No easy re-consent
Disengaged participants make it impossible to update permissions for new studies.
Disconnected metadata
Samples lack attached phenotype and genotype data needed for feasibility analysis.
Secondary use authorization always current labeled as authorized, restricted, or expiring
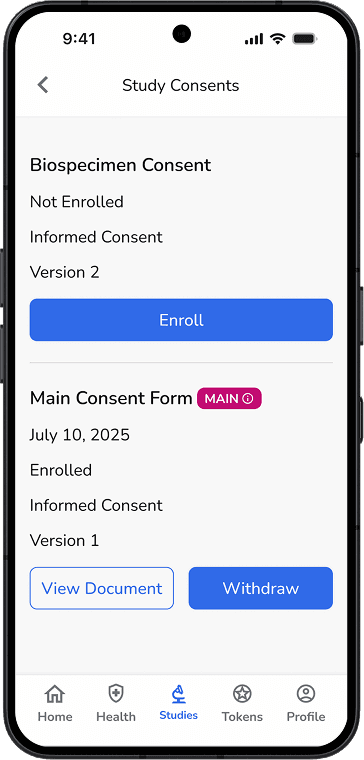
Re-consent when needed via secure participant outreach
Clinical + sample metadata unified for instant filtering & feasibility
Participants can enroll, update, or withdraw consent
Identity verification & audit trail
Permissions for secondary and ancillary studies